RegTech
Regulation Technologies for Medical Devices
Medical device development processes increasingly involve more data, documentation, and regulatory requirements.
For this reason, traditional document-based regulatory management is no longer sufficient.
The RegTech approach aims to manage regulatory processes through digital platforms. With this approach:
- All requirements can be tracked,
- Risk and testing processes can be linked,
- Technical file processes can be kept up to date.
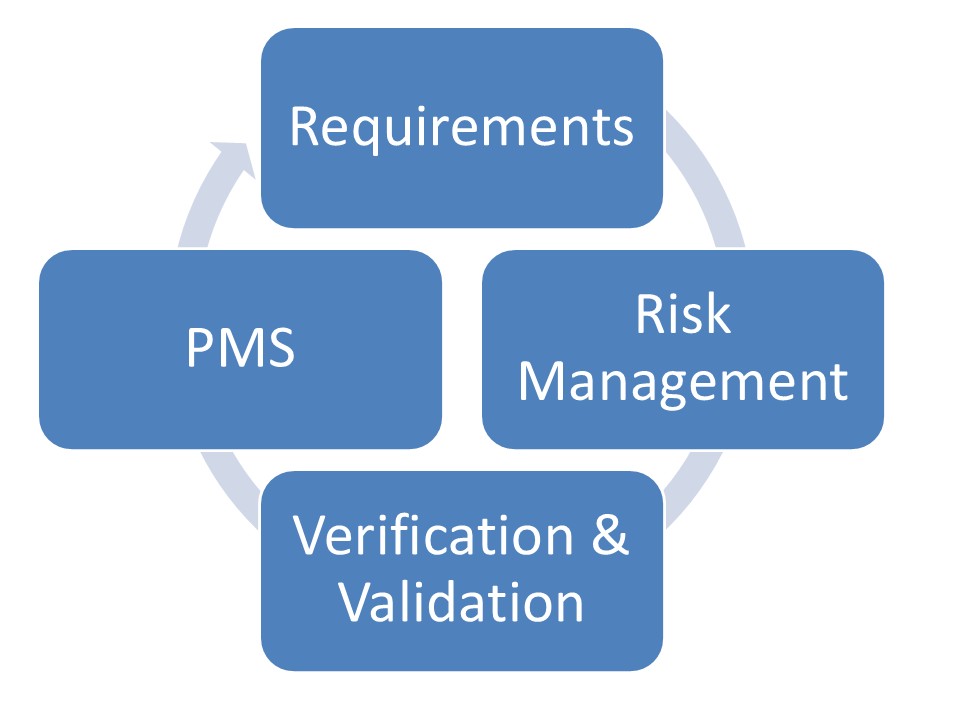
Closed-Loop Regulatory Lifecycle
Modern regulatory management is structured around the following cycle: Thanks to this closed-loop approach, regulatory compliance can be maintained throughout the product lifecycle.
- Requirements,
- Risk Management,
- Verification & Validation,
- Post-Market Surveillance (PMS).

Orcanos Platform
Orcanos is a RegTech platform that integrates requirements management, risk management, and test management into medical device development processes. With Orcanos, you can: With Orcanos:
- Design review processes become digital,
- Technical document management becomes easier,
- MDR compliance processes are accelerated.
For requests for brochures and demos info@coruhty.com

